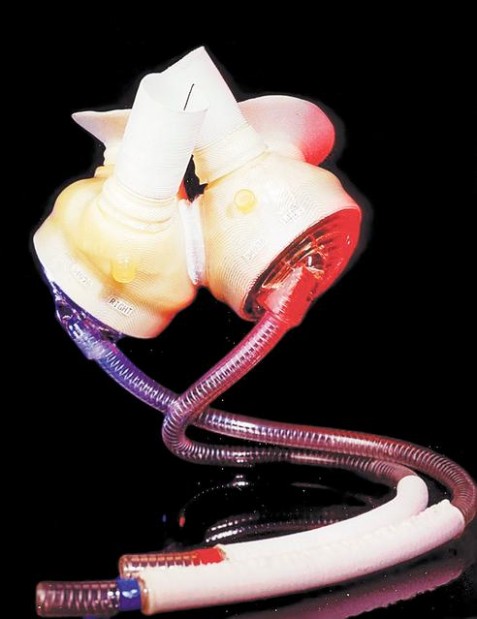
Since becoming the first artificial heart to win federal approval for commercial use last year, the CardioWest total artificial heart has been a medical success.
Now, new management at CardioWest maker SynCardia Systems Inc. of Tucson is looking to translate that clinical success to the marketplace.
By February, SynCardia plans to submit for Food and Drug Administration approval a new pneumatic driver, or pump, to power the CardioWest, company officials said.
Earlier this year, two investors assumed key posts at SynCardia, a privately held company that employs 24 people.
Rodger Ford, an entrepreneur and investor who founded AlphaGraphics Printshops of the Future, quietly assumed the post of SynCardia's chief executive officer in May. Also joining the company was David Mackstaller, a co-founder with Ford of Anthem Equity Group Inc., a Tucson real estate and business development company and a key SynCardia investor.
People are also reading…
Mackstaller is "heart center selection director" for SynCardia, a post in which he is identifying heart-transplant centers as possible customers for the CardioWest heart.
Evolved from the first artificial heart implanted in a human — the 1980s-era Jarvik-7 — the CardioWest heart is implanted in patients to keep them alive until they receive human donor hearts.
The CardioWest and its earlier versions have been used in about 550 patients since 1985, when a University Medical Center team led by University of Arizona heart surgeon Dr. Jack Copeland first implanted the heart to keep a patient alive until a donor heart transplant, Mackstaller said.
In August, the heart was used for the first time commercially — by a hospital that had not been previously involved in research on the device — when the Cleveland Clinic implanted the device in a dying man in his 40s.
Now, the new management team is looking to take SynCardia to the next level as a business, Ford said.
"This company has been a 25-year science project, pretty much," he said.
"The heart hasn't been in wide distribution, so this company hasn't had business systems, it's had development systems."
Ford said SynCardia has 10 heart-center customers, including UMC and the Cleveland Clinic in the United States, two hospitals in France, two in Germany and one in Canada.
The company's goal is to serve most of the 200 hospitals worldwide that perform heart transplants annually and can meet stringent FDA and company criteria to be eligible to use the product, Mackstaller said.
The new pneumatic driver will be a key to that marketing plan, Mackstaller and Ford said.
In the past year, SynCardia has sold about 60 CardioWest hearts, at about $100,000 each, Mackstaller said.
But the company is still using the same 37 pneumatic "drivers" inherited from Jarvik's company, Symbion, to power the CardioWest hearts.
"All of a sudden, we had an FDA-approved heart with no way to drive it except for technology that is 20 years old," Ford said.
The company has spent the last four years acquiring new, portable driver technology for the CardioWest from companies in Europe and recently licensed technology for a mobile driver unit from MEDOS Medizintechnik AG, a German company.
SynCardia recently began reliability testing of the new drive system at a Houston laboratory, as part of efforts to gain U.S. and European regulatory approvals for the new system.
The company plans to submit the system for FDA approval by mid-February and win the agency's blessing later next year.
"We are proud that the product of SynCardia is a life-saving device, but also proud that a product of the company is the company itself," Dr. Marvin J. Slepian, SynCardia co-founder, chairman and chief scientific officer, said in a statement announcing the new management team this week.
Slepian is professor of cardiology, director of interventional cardiology, and director of the Tissue Engineering Lab at the UA.
Richard G. Smith, director of the artificial-heart program at UMC's Sarver Heart Center, is SynCardia's co-founder and chief technical officer.
TECH FILE